New Research Shows Bacteria Essential To Health
How Your Own Bacteria Help Prevent Disease
(Continued)
Health, Disease And The State Of Your Microbiome
Scientists are discovering numerous potential links between an off-balance microbiome and disease. It seems our microbiome significantly enriches our digestion; in fact humans can be described as super-organisms whose metabolism represents an amalgamation of microbial and human attributes.
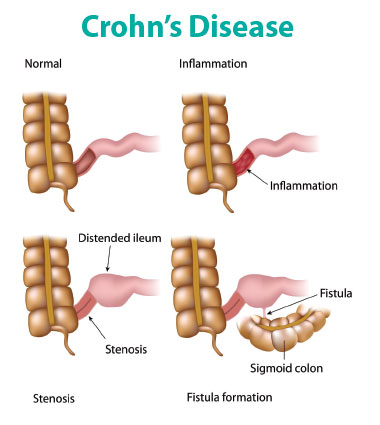 |
| Crohn’s Disease
Click to enlarge |
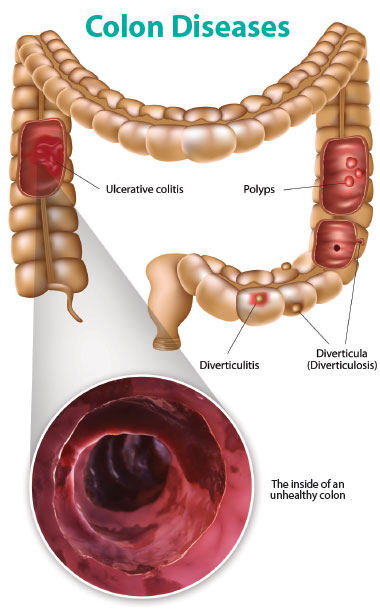 |
| Colon Diseases
Click to enlarge |
For example, differences in the proportions of two types of gut bacteria even suggest a correlation with obesity. Obese people have more of one group of bacteria, Firmicutes, and fewer of another group, Bacteroidetes, than thin ones. When weight is lost, the balance of Firmicutes and Bacteroidetes changes to match. Bacteroidetes appear to suppress both the production of a hormone involved in storing fat and an enzyme that prevents fat from being burned. On the other end of the spectrum microbes in the guts of malnourished people are unable to synthesize vitamins and digest certain nutrients.
The ability of the gut microbiome to alter our metabolism may result in metabolic (bodily) and/or immune (defense system) regulation changes. Autoimmune disease, in which the body’s defense mechanism fails to be protective (diabetes and rheumatoid arthritis, for example) may begin as an immune system attack on gut bacteria. The gut has a mucosal barrier to prevent organisms from crossing from the digestive tract into the body. If bacteria that normally would be recognized as “good” breach the barrier, they sometimes trigger an autoimmune response in which the body attacks itself. Additionally, significant changes have been observed in the gut microbe populations of people with various types of Inflammatory Bowell Disease (IBD), such as Crohn’s disease or colitis, including a loss of normally protective species, when compared with bacteria in healthy people.
Lifestyle Changes and Choices
Our microbiome is also providing more evidence for the adverse effects of certain lifestyle choices. Take the modern Western diet. Historical dietary shifts have influenced the oral microbiome: The frequency of Streptococcus mutans, the bacteria most associated with caries (tooth decay), is significantly higher in modern biofilm samples than in preindustrial ones. This indicates that these bacteria have only become dominant since medieval times, and with the onset of agriculture.
In the oral cavity too, cigarette smoking makes the environment more hospitable for organisms associated with periodontal (gum) disease. After a person stops smoking, however, the mouth and upper respiratory tract are recolonized with a greater population of health-promoting microbes and fewer pathogens.
The composition of the oral microbiome is dominated by 5 phyla (groupings) of bacteria that account for over 95% of its roughly 10,000 bacterial species. Oral structures and tissues (such as the tongue, cheeks and teeth) are colonized by distinct microbial communities.
Periodontal (gum) disease has traditionally been associated with the development of specific bacterial complexes in the sub-gingival (“sub” – beneath; “gingival” – gum) biofilm. Now, cutting-edge technology is allowing scientists to reconstruct the genomes of bacteria that cannot be cultured in laboratory settings, thus increasing understanding of their potential role in this destructive disease that destroys the supporting structures of the teeth.
New Treatment Possibilities
When harmful bacteria threaten, the customary response has been “broad spectrum” antibiotic medication. A problem with this “sledgehammer to crack a nut” approach is that this medication kills the good bacteria along with the bad, destroying the normal balance. Furthermore certain pathogenic (disease-causing) species have developed resistance to many customarily used antibiotics.
One example is Clostridium difficile, which can cause symptoms ranging from diarrhea to life-threatening inflammation of the colon. Another is methicillin-resistant Staphylococcus aureus (MRSA), a variant of a germ commonly found on our skin and in our noses. Occasionally it gets past the skin barrier, usually through an injury, and causes a minor skin infection. The MRSA strain does not respond to the usual arsenal of antibiotics that prevent the infection from spreading to the bloodstream and other areas of the body and can be rapidly fatal.
 |
| MRSA Superbugs. |
With new knowledge about our microbiome and how it works, scientists are excited about the possibilities of restoring health by rebalancing a sick person’s microbial mix, and preventing disease by nurturing the healthy microbiome. A promising therapy already in use by some doctors is fecal transplant from a healthy donor to treat Clostridium difficile. Although a diet of probiotic yogurt might seem like a more pleasant alternative, a study published in 2011 showed its effects on gut bacteria are much more subtle.
We are born 100% human and then live and die as a “super-organism” that contains 10 times more microbial cells than human cells and 100 times more microbial genes than human ones! We are the sum of our parts, and clearly one of those parts is our microbiome — without which we cannot live normal, healthy lives.

